The North American Consensus on Breath Testing recommends using a hydrogen and methane breath test to detect Small Intestinal Bacterial Overgrowth (SIBO), abnormal growth of bacteria in a patient’s small intestine, and Intestinal Methanogen Overgrowth (IMO), excessive levels of methane in a patient’s intestinal tract. Both SIBO and IMO can cause various symptoms in patients, including diarrhea, nausea, bloating, gas, and abdominal cramps.
To help healthcare providers get started with ordering breath tests for their patients, CDI’s provider support team has compiled six of the most frequently asked questions:
What is a hydrogen & methane breath test?
 A hydrogen and methane breath test is a non-invasive diagnostic tool that can be administered in-office or in the comfort of the patient’s own home. During the hydrogen and methane breath test for SIBO and IMO, the presence of elevated levels of hydrogen or methane gas, or both, identified within 90 minutes of ingesting a substrate solution provides evidence of bacteria or methanogens in the upper region of the digestive tract, i.e., at the level of the small intestine.
A hydrogen and methane breath test is a non-invasive diagnostic tool that can be administered in-office or in the comfort of the patient’s own home. During the hydrogen and methane breath test for SIBO and IMO, the presence of elevated levels of hydrogen or methane gas, or both, identified within 90 minutes of ingesting a substrate solution provides evidence of bacteria or methanogens in the upper region of the digestive tract, i.e., at the level of the small intestine.
This excessive build-up of bacteria is often diagnosed as SIBO, while excessive methanogens that cause methane gas are often interpreted as IMO.
What is the difference between glucose and lactulose substrates?
Since the digestive tract absorbs glucose, it will not detect SIBO further along in the small intestine (proximal jejunum/ileum). However, glucose is more sensitive than lactulose, providing fewer false positives with colonic bacteria.
On the other hand, the digestive tract does not absorb lactulose, so it will travel through the entire gastrointestinal tract, ultimately providing a complete depiction of all intestinal segments, including the colon (large intestine).
Providers can choose between the two substrates when ordering a test for patients.
NOTE ON ALLERGIES: Most substrates do not come with food allergy labeling, so selecting the appropriate substrate is essential when ordering a test kit for a patient.
Are there any risks associated with this test?
There are no risks involved in performing a hydrogen and methane breath test. Patients may experience some of their usual symptoms during the test, which is normal.
In light of COVID-19, there are significant benefits to at-home breath testing due to aerosols’ effects in spreading the virus. Giving patients the ability to take a diagnostic test at home mitigates the risk of exposure for both the patient and healthcare staff.
What preparation is required for a patient to take a test?
Patients must follow the required test prep instructions to ensure accurate results.
Test restrictions: Patients should discontinue the use of any antibiotics for four weeks before taking the test, discontinue the use of any laxatives and/or promotility drugs (i.e., anything that helps aid in the movement of your bowels) for one week before taking the test, not smoke or vape for at least 24 hours before the test, or any time during the test, and not sleep or exercise for at least one hour before taking the test or at any time during the test.
A 24-hour diet preparation period, which includes a 12-hour limited diet and a 12-hour fasting period, is also required before taking a test (see “Preparing for Sample Collection” section of Instructions for Use). It may be convenient for a patient to begin the 24-hour preparation period in the morning to follow the specific diet during the day, immediately followed by the fasting period during the night (which may include time spent sleeping).
Patients with diabetes should not use this test due to the amount of absorbable sugar and fasting required.
What happens during the test?
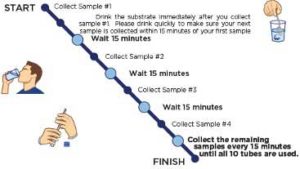 The patient should have followed the 12-hour limited diet and have fasted for the 12 hours before starting the test. The patient will mix the test substrate (lactulose or glucose) with water and set it aside. The patient will give a baseline breath test sample when the test begins by blowing into a test tube with the provided straw. The patient will then drink the substrate mixture immediately following the collection of the baseline sample. After drinking the substrate, the patient will give a breath sample every 15 minutes starting from when they took their first baseline sample for 2 hours and 15 minutes. Patients should not eat, chew gum, smoke, sleep or exercise during the test.
The patient should have followed the 12-hour limited diet and have fasted for the 12 hours before starting the test. The patient will mix the test substrate (lactulose or glucose) with water and set it aside. The patient will give a baseline breath test sample when the test begins by blowing into a test tube with the provided straw. The patient will then drink the substrate mixture immediately following the collection of the baseline sample. After drinking the substrate, the patient will give a breath sample every 15 minutes starting from when they took their first baseline sample for 2 hours and 15 minutes. Patients should not eat, chew gum, smoke, sleep or exercise during the test.
The test will last 2 hours and 15 minutes. After the test, patients are free to return to their usual diet, medication, and daily activity.
How will providers and patients get their results?
 Patients are responsible for shipping their completed test kit to CDI’s CLIA-certified laboratory using the pre-paid shipping label provided in the test kit. CDI’s lab must receive a patient’s samples within two weeks of collection. Sending the kit within 24 hours of taking the test is recommended.
Patients are responsible for shipping their completed test kit to CDI’s CLIA-certified laboratory using the pre-paid shipping label provided in the test kit. CDI’s lab must receive a patient’s samples within two weeks of collection. Sending the kit within 24 hours of taking the test is recommended.
Once CDI receives the patient’s test kit, it is typically processed and analyzed within one business day. Your practice will receive results via a secure fax system, secure email, or your office can set up an account with CDI to obtain results via our HIPAA-compliant web-based reporting system.
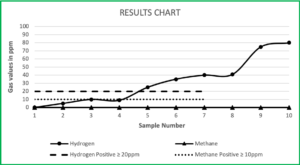
Healthcare providers are responsible for reviewing test results with their patients. CDI provides a list of the six most common results to assist with understanding the data presented. CDI offers complimentary test result interpretation at the request of the healthcare provider.
CDI’s hydrogen and methane breath test kits are available to order for patients by healthcare providers at no cost and are easily accessible through our online ordering portal. Additional educational materials, instructional guides, and FAQs are also available in our online Provider Resource Center. Contact CDI’s dedicated provider support team at 1-888-258-5966 or customerservice@commdx.com with any additional questions.

